R-Evolution WorldWide, has recently submitted the project proposal IMPROVE-HIV under the European & Developing Countries Clinical Trials Partnership (EDCTP) call HORIZON-JU-GH-EDCTP3-2026-01-HIV-03-two-stage – Global collaboration action towards better prevention, treatment and clinical management of HIV coinfections or co-morbidities in sub-Saharan Africa.
The proposal was developed by an international consortium including R-EvoWW, Queen Mary University of London (United Kingdom), the Infectious Diseases Institute – Makerere University (Uganda), and the Tropical Gastroenterology and Nutrition Group (Zambia), bringing together expertise in HIV clinical research, global health, hepatology, parasitic diseases, and implementation science.
Despite the large-scale expansion of antiretroviral therapy across sub-Saharan Africa, HIV-associated co-infections and co-morbidities remain major drivers of preventable morbidity and health-service burden. Chronic hepatitis B virus (HBV) infection and schistosomiasis are highly prevalent among people living with HIV in countries such as Zambia and Uganda, where they significantly increase the risk of liver inflammation, fibrosis, and treatment complexity.
IMPROVE-HIV proposes a pragmatic Phase III randomised, double-blind, placebo-controlled effectiveness trial embedded within routine HIV services (see figure below). The study will evaluate whether adding fixed-dose multiple micronutrient supplementation to the standard of care can reduce immune activation and liver injury in individuals living with HIV and HBV, schistosomiasis, or both. The project will assess clinically relevant outcomes using biomarkers already integrated in routine monitoring, together with indicators of immune recovery, treatment continuity, nutritional status, and safety.
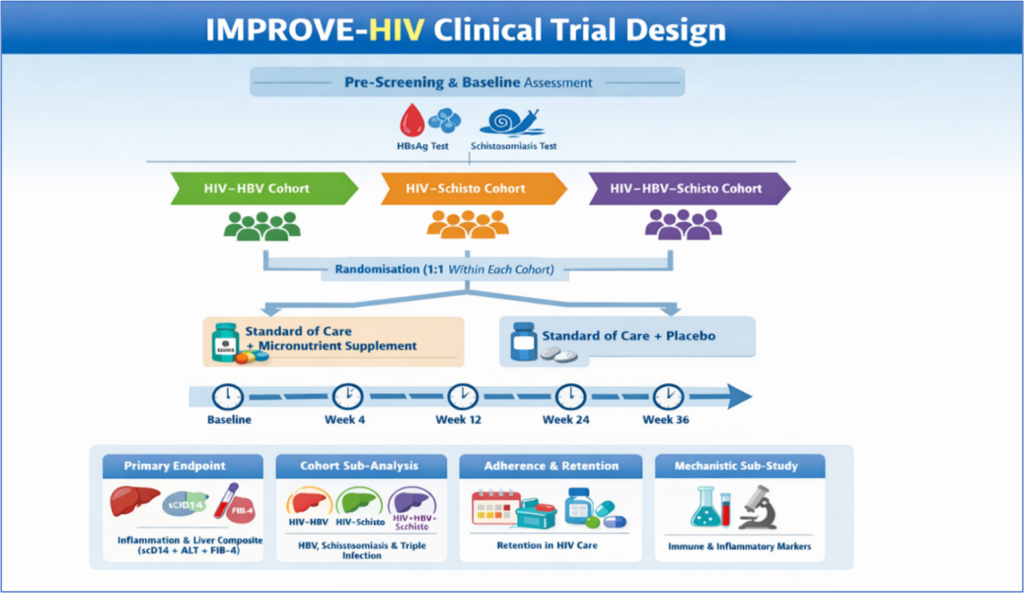
In addition to the clinical trial, the project includes cost-effectiveness and implementation research components, with the aim of generating decision-ready evidence that can support national HIV and hepatitis programmes and facilitate the rapid uptake of effective, scalable interventions.
Through this proposal, the consortium aims to contribute to the development of practical and sustainable strategies for the integrated management of HIV and its co-morbidities, in line with EDCTP priorities and with the broader goal of strengthening health systems in sub-Saharan Africa.



