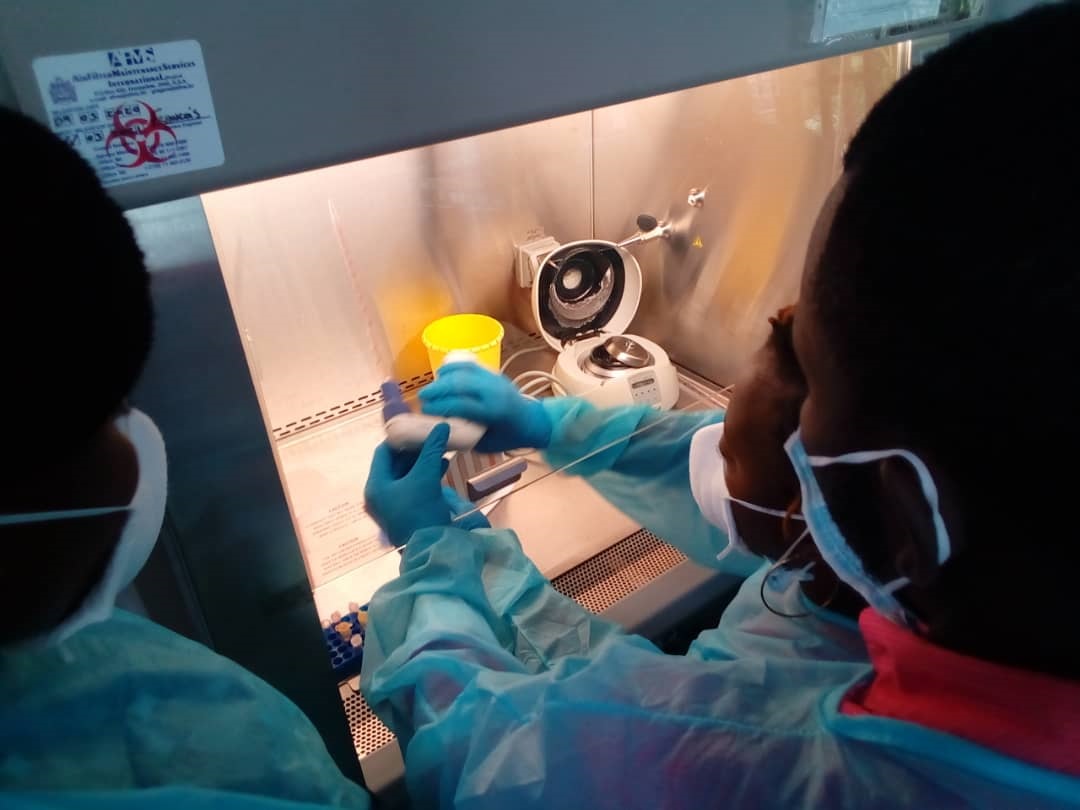
As part of the strengthening of health systems and in particular the regulation of drugs and health products within the Africlinique project, the headquarters of the World Health Organization (WHO), the WHO Regional Office for Africa (AFRO), and the WHO Country Office (PO) in the Republic of Congo provided technical assistance to support the Directorate of Pharmacy and Medicine (DPM) of Congo from February 15 to 19 2021 in Brazzaville (Republic of Congo).
The Global Benchmarking Tool (GBT) integrates the Maturity Level (ML) of the various drug regulatory functions in a computerized platform that facilitates its use and the ranking of maturity indicators. This self-assessment has enabled several countries in the sub-region to identify their ML as well as to put in place institutional development plans (IDPs) in order to access a ML which guarantees safety, quality and effectiveness of drugs in development and in circulation.
Click here to for news release in English.